|
1/12/2024 0 Comments No2 molecular geometryHydrogen has an electronic configuration of 1s 1. 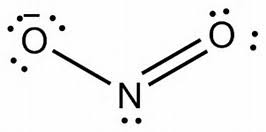
Therefore, the lone Nitrogen atom contributes 5 x 1 = 5 valence electrons. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3. Nitrous acid comprises two Oxygen atoms, one Hydrogen atom, and one nitrogen atom. It has the following properties: Name of the molecule It also reacts with aliphatic alcohols to produce alkyl nitrites which act potently to counteract Vasoconstriction. Nitrous acid also finds use as a precursor in the production of Adipic acid, which is a key component in the production of Nylon. This simple synthesis is the crucial step in the production of commercial dyes. HNO 2 reacts with amines to form diazonium salts, which play a vital role in the production of azo dyes. Nitrous acid plays an important role in the production of diazonium salts. It is mainly used as a preservative in the processed meats industry. One of the more prominent Nitrites (salts of Nitrous acid) is NaNO 2 (Sodium Nitrite). Again, due to its unstable nature, it can react as either an oxidizing or reducing agent. Nitrous acid decomposes into Nitric Oxide (NO) and Nitric Acid (HNO 3 ) at elevated temperatures. Its existence in these peculiar states is due to the fact that HNO 2 is unstable and decomposes rapidly in its free form. It appears to be pale blue as a solution. 
It is a weak acid and exists only in specific conditions, i.e., in solution (cold and dilute), as a gas, or in the form of nitrite salts. HNO 2 is also known as Dioxonitric(III) acid. The chemical formula HNO 2 represents Nitrous Acid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
